

Hexanes are highly flammable solvents derived from petroleum, commonly used in laboratories and industrial extraction. They are a group of six-carbon hydrocarbons valued for their strong dissolving power and rapid evaporation. The primary dangers of hexane involve fire risk and nervous system damage from prolonged exposure. Safe handling depends on ventilation, exposure monitoring, and proper chemical management systems.
This guide explains the science, the health risks, and the control measures for Hexane.
Physical and Chemical Properties of Hexane
Hexane refers to several structural isomers with the formula C₆H₁₄. In industrial settings, the term usually describes a mixture, often rich in n-hexane.
Volatility and Odor
Hexane evaporates quickly at room temperature. It has a mild, petroleum-like odor. However, odor is not a reliable safety indicator. Concentrations can reach hazardous levels before the smell becomes obvious.
Its boiling point is approximately 69°C (for n-hexane), which contributes to rapid vapor formation. Vapors are heavier than air and can travel to ignition sources.
Solubility and Chemical Behavior
Hexanes mix easily with oily and greasy substances, such as fats, waxes, and many industrial chemicals. It does not mix with water. In simple terms, it dissolves oil-based materials but separates from anything water-based.
This non-polarity explains its widespread use in:
- Oilseed extraction
- Adhesive formulation
- Cleaning and degreasing
- Laboratory chromatography
Because hexane does not mix with water, spills can spread across surfaces and evaporate quickly, increasing fire and inhalation risk.
Understanding the Dangers of Hexane Exposure
The dangers of hexane extend beyond flammability. Health effects depend on exposure levels and duration.
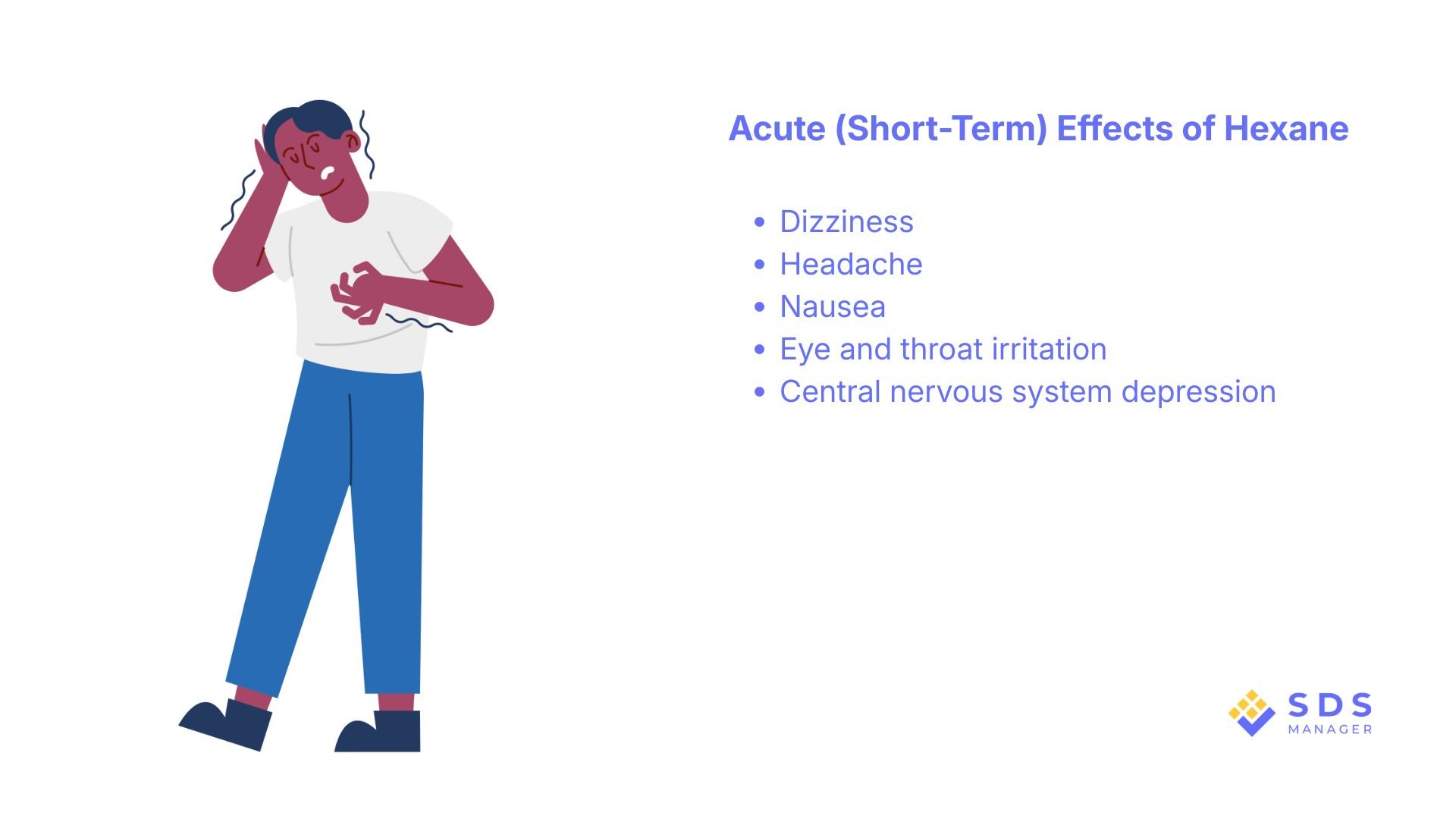
Acute (Short-Term) Effects
Short-term inhalation may cause:
- Dizziness
- Headache
- Nausea
- Eye and throat irritation
- Central nervous system depression
High vapor concentrations can lead to confusion or unconsciousness in poorly ventilated areas.
Chronic (Long-Term) Effects
Prolonged exposure to n-hexane can damage peripheral nerves. Workers may develop numbness, tingling, or muscle weakness. This condition is called peripheral neuropathy.
The toxic mechanism involves a metabolite known as 2,5-hexanedione. It interferes with nerve protein structure. This is why long-term occupational monitoring is important.
To reduce these risks, the Occupational Safety and Health Administration (OSHA) enforces a Permissible Exposure Limit (PEL) of 500 ppm over an 8-hour shift for n-hexane. Employers must monitor airborne concentrations and implement engineering controls when necessary.
Hexane can catch fire very easily. It gives off flammable vapors even at very low temperatures, and a small spark, including static electricity, can ignite those vapors.
How to Safely Store and Handle Hexane
Safe storage and handling depend on the environment and volume.
Ventilation Requirements
Always use hexane in well-ventilated areas. In laboratories, a chemical fume hood is essential. In industrial facilities, local exhaust ventilation prevents vapor buildup.
Confined spaces require atmospheric testing before entry. Vapor accumulation in enclosed areas increases both toxicity and explosion risk.
While physical ventilation is key, the first step in risk control is maintaining an online safety data sheet inventory to ensure every worker has instant access to the latest hexane safety data.
Grounding and Bonding
Because hexane is highly flammable, static electricity must be controlled. Ground and bond containers during transfer. Use explosion-proof equipment in classified areas.
Secondary Containment
Store hexane in approved flammable liquid cabinets. Use secondary containment to prevent environmental release. Keep containers tightly closed when not in use.
For organizations managing multiple hazardous substances, maintaining an accurate safety data sheet through a centralized chemical management platform improves compliance and audit readiness.
Selecting PPE for Hexane Protection
You might reach for standard nitrile gloves out of habit, but they offer almost zero protection here. Hexane can permeate nitrile in just minutes, drenching your skin underneath the glove.
For real protection, you must use materials like Viton or Silver Shield. These laminates provide the necessary barrier against solvent breakthrough. Selecting the correct PPE is a core requirement of OSHA’s Hazard Communication Standard.
Hexane Spill Response and Disposal
If a leak occurs, your first 60 seconds are critical. Evacuate the area and eliminate all ignition sources immediately. For small spills, use non-combustible absorbents like vermiculite. To ensure your team is prepared for such an event, you can download our Chemical Spill Response Procedure and Safety Guidelines.
In confined spaces, the flammability risk is magnified. You must use spark-proof tools for cleanup, as a single scrape of metal on concrete could ignite the concentrated vapors. Once you stabilize the liquid, it must be moved to a secure, labeled container for transport. Because hexane is a listed hazardous waste (EPA Code D001), disposal must follow RCRA guidelines to avoid environmental contamination. Never pour it down the drain as the vapors can linger in plumbing and cause explosions.
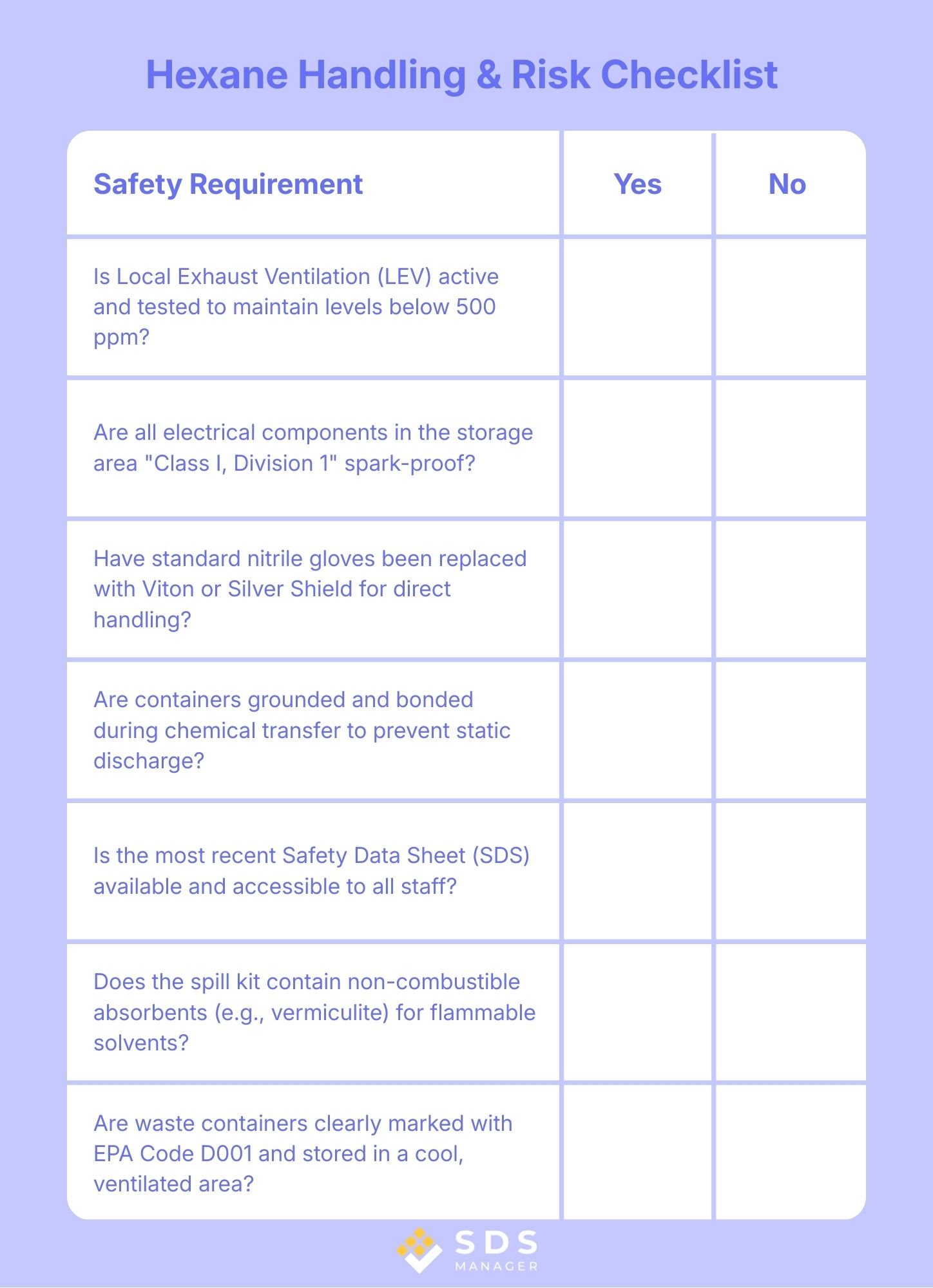
Hexane Handling & Risk Checklist
Use this checklist to ensure your facility meets standard industrial safety protocols.
- Vapor Control: Is local exhaust ventilation (LEV) active and tested to maintain levels below 500 ppm?
- Ignition Prevention: Are all electrical components in the storage area "Class I, Division 1" spark-proof?
- PPE Audit: Have standard nitrile gloves been replaced with Viton or Silver Shield for direct handling?
- Static Grounding: Are containers grounded and bonded during chemical transfer to prevent static discharge?
- SDS Access: Is the most recent safety data sheet available?
- Spill Kit Readiness: Does your spill kit contain non-combustible absorbents (like vermiculite) specifically for flammable solvents?
- Waste Marking: Are waste containers clearly marked with EPA Code D001 and stored in a cool, ventilated area?
You can download the checklist image below to print and post in your chemical handling and storage areas for daily reference.
Final Takeaway
Hexane is efficient and versatile, but it demands disciplined control. The dangers of hexane arise from volatility, neurotoxicity, and extreme flammability.
Safe use depends on engineering controls first, PPE second, and documentation always.
Control the environment. Control the exposure. Control the outcome.
Frequently Asked Questions
Is hexane the same as n-hexane?
Not exactly. “Hexane” often refers to a mixture of isomers. n-Hexane is a specific straight-chain form with higher neurotoxicity concern.
Does hexane evaporate quickly?
Yes. It has high volatility and forms vapors at room temperature.
Is hexane flammable?
Extremely. It has a very low flash point and wide explosive limits.
Can hexane exposure cause nerve damage?
Yes. Chronic inhalation of hexane can lead to peripheral neuropathy.
Where can I access reliable safety data for hexane?
Maintain up-to-date documentation through a structured chemical safety data management system that centralizes Safety Data Sheets and compliance records.

