

ECHA inspected 2,528 Safety Data Sheets across 28 EU and EEA countries as part of its REF-11 enforcement project. One in three failed.
The findings, published in the official REF-11 project report, cover failures in classification, nanoform disclosures, endocrine-disrupting property data, and exposure scenarios. With CLP new hazard class deadlines arriving in November 2026 and ECHA's follow-up enforcement projects already underway, the pressure on SDS quality across the EU supply chain is higher than it has ever been.
If your SDS library has not been reviewed against the updated REACH Annex II requirements (EU Regulation 2020/878), the findings below are worth reading carefully.
WHAT ECHA FOUND
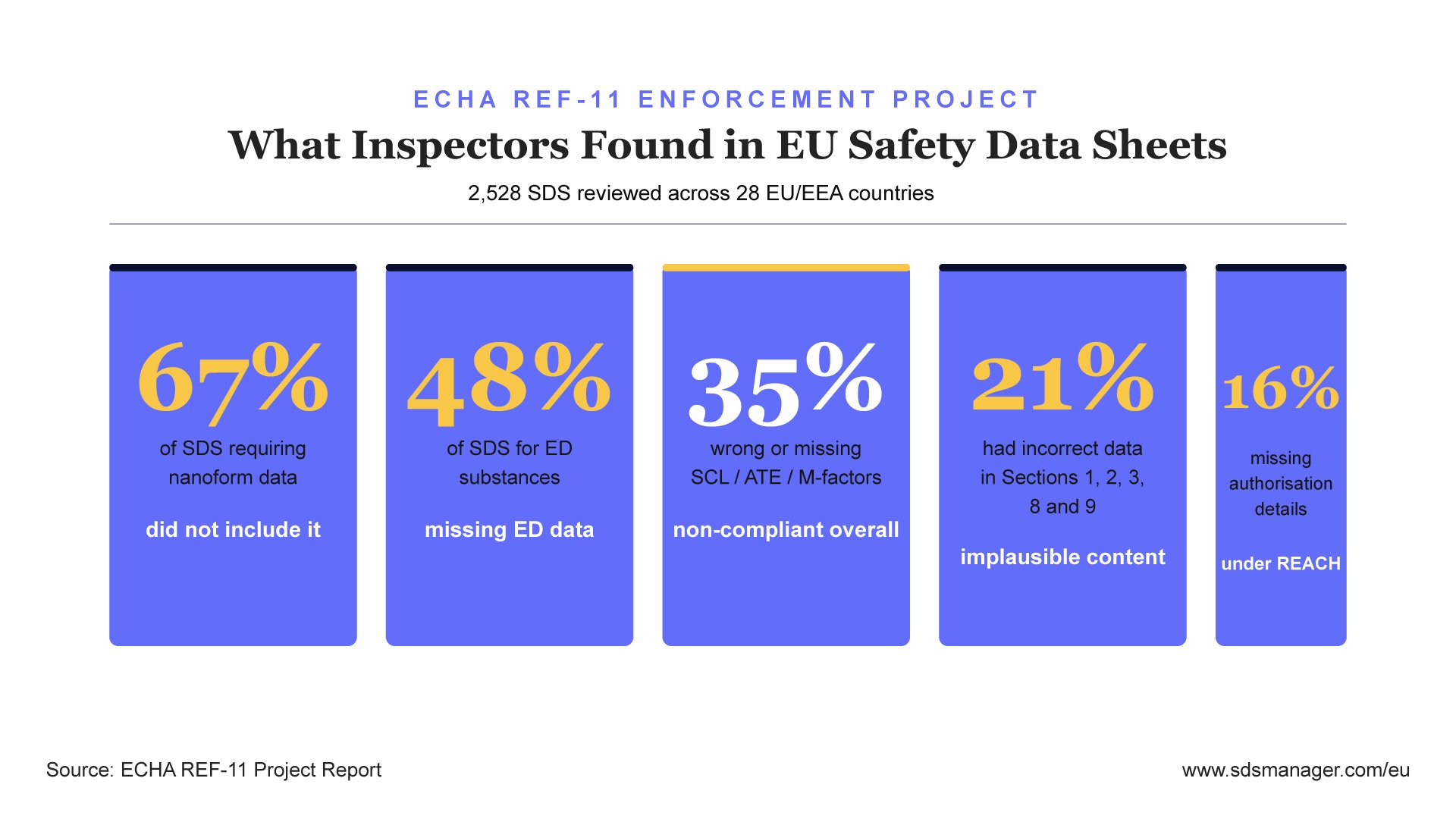
The REF-11 project examined 2,528 SDS across 28 countries and found that 35 % did not comply with the legislation. The non-compliance was not concentrated in one or two sections. Inspectors found failures throughout the document, from the first page to the physical and chemical properties sections.
The breakdown by failure type:
- 67 % of SDS that required nanoform information did not include it
- 35 % of SDS for substances with endocrine-disrupting properties were missing the required data
- 35 % of SDS had incorrect or missing specific concentration limits (SCL), acute toxicity estimates (ATE), or multiplication factors (M-factors)
- 21 % had incorrect or implausible data in Sections 1, 2, 3, 8, and 9
- 16 % were missing required authorisation details under REACH
Even among companies that had active procedures to update and distribute SDS (81 % of those inspected did), one in three documents still failed on content quality.
Source: ECHA REF-11 Project Report, December 2024
THE MOST COMMON FAILURES, SECTION BY SECTION
Inspectors flagged specific gaps in the following sections:
Section 1 — UFI codes missing or placed in the wrong subsection (should appear in 1.1, not 2.2)
Section 2 — Missing authorisation numbers in subsection 2.2. Missing endocrine-disrupting property information in subsection 2.3. Incorrect classification and labelling in subsections 2.1 and 2.2
Section 3 — Missing specific concentration limits (SCLs), M-factors, and ATEs. Incorrect ingredient classification
Section 8 — Missing occupational exposure limit (OEL) values, required notations, and legal bases in subsection 8.1. Incomplete PPE information in subsection 8.2, including missing glove thickness, applicable standards, and permeability times
Section 9 — Missing or incomplete data on nanoforms. Omission of available physical and chemical property data
These are not edge cases or obscure requirements. Most of them stem directly from Regulation (EU) 2020/878, which updated REACH Annex II and has been in force since January 2021.

WHAT HAPPENS WHEN AN SDS FAILS INSPECTION
Enforcement under REF-11 followed a structured escalation. Of the 903 non-compliant SDS identified:
- 11 % received written advice from national authorities
- 11 % received administrative orders requiring corrective action
- 11 % were issued fines
- 6 % resulted in criminal complaints
Written advice is the starting point, not the ceiling. Companies that do not act on written recommendations move up the escalation ladder. At the national level, this can include administrative penalties, market access restrictions, and in serious cases, product withdrawal from the EU market.
In 2026, ECHA's compliance check activity is intensifying. The agency conducted 313 REACH compliance checks in the past year, issuing 208 data requests and referring 30 % of non-responding companies to national enforcement authorities.

Source: ECHA Enforcement Forum
WHY 2026 MAKES THIS MORE URGENT
REF-11 assessed compliance with requirements that have been in place since 2021. The regulatory picture in 2026 is more demanding, not less.
November 1, 2026: The new CLP hazard classes for substances become mandatory. Endocrine disruptors (ED), PBT/vPvB, and PMT/vPvM classifications must be reflected in SDS for substances already on the market before May 2025.
May 1, 2026: The same new hazard classes apply to mixtures placed on the market for the first time. Companies must already be assessing their formulations now.
REF-12 and REF-13: ECHA's next two enforcement projects are already running. REF-12 targets imported chemicals and whether their documentation meets EU requirements at the border. REF-13 targets online chemical sales. Both projects feed findings to national enforcement authorities.
Companies that failed REF-11 requirements are now operating in a regulatory environment where ECHA has more tools, more data, and more cross-border coordination than at any point before.

WHAT TO CHECK IN YOUR SDS LIBRARY RIGHT NOW
Based on the REF-11 findings, three areas warrant immediate review:
- Nanoform disclosures. If any substance in your product formulations exists in nanoform, your SDS must include specific particle characterisation data. This was the single highest failure rate in the entire project at 67 % . Check every SDS where nanoform variants are relevant to your supply chain.
- Endocrine-disrupting property data. Subsection 2.3 must now include a statement on endocrine-disrupting properties where applicable. With the new CLP hazard classes making ED classification mandatory before November 2026, any SDS missing this information is already behind.
- Section 8 PPE and OEL completeness. Inspectors repeatedly found incomplete PPE data, missing glove permeability times and applicable standards, and absent OEL values with their legal bases. Section 8 is one of the first sections a workplace inspector checks. Gaps here are both a REACH compliance failure and a worker safety issue.

READY TO REVIEW YOUR EU SDS LIBRARY?
SDS Manager gives EU compliance teams centralised access to their full SDS library, with automated update tracking and audit-ready documentation across all sites.

