

You probably use acetic acid every time you dress a salad or clean a surface at home. Most people just call it vinegar, not realizing it's the same chemical that, at higher concentrations, can cause serious burns and permanent eye damage.
Acetic acid is a colorless, sharp-smelling acid best known as the active ingredient in vinegar. At low concentrations, it's mostly harmless. At high concentrations, it's a chemical hazard that needs to be handled carefully.
This article covers what acetic acid is, where it's found, what it's used for, the real dangers it poses, and how to stay safe around it.
What Is Acetic Acid?
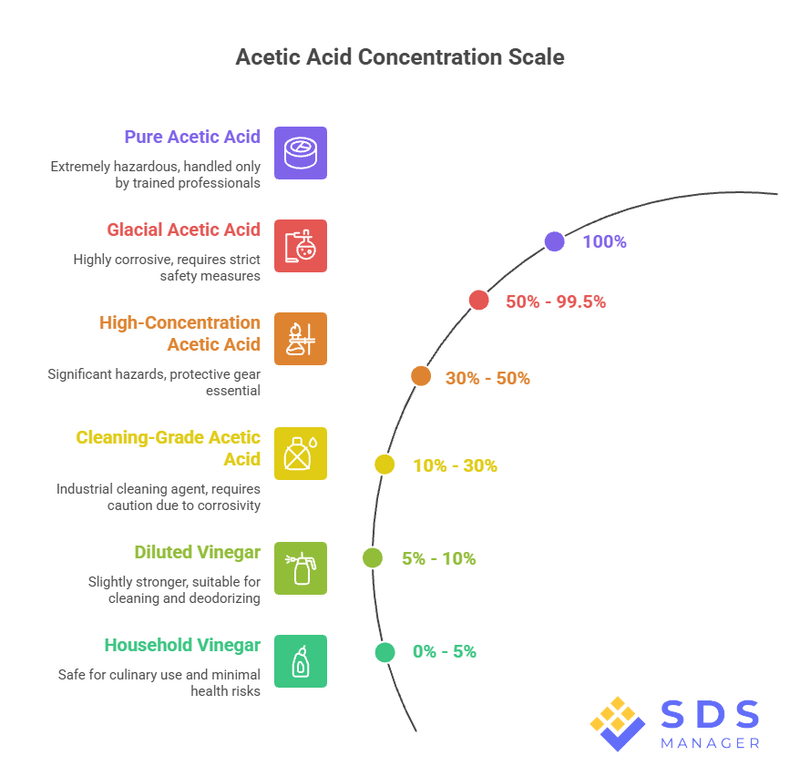
Acetic acid (chemical formula CH₃COOH) is also called ethanoic acid. It's a weak acid that gives vinegar its sour taste and sharp smell. Regular household vinegar contains about 5 to 8% acetic acid, with the rest being water.
In its pure form, it's called glacial acetic acid, a colorless, corrosive liquid with a concentration of 99.5% or higher. It actually solidifies below 16.7°C (62°F), which is where the "glacial" name comes from. This form is far more dangerous than the vinegar in your kitchen.
Acetic acid has been used for thousands of years. It forms naturally when certain bacteria break down alcohol in liquids like wine or apple cider, which is essentially how vinegar has always been made.
Where Is Acetic Acid Naturally Found?
Acetic acid forms naturally during fermentation. When Acetobacter bacteria break down the alcohol in liquids like wine or cider, the result is acetic acid. That's the basic process behind making vinegar.
You can also find small amounts of it in:
- Ripening fruit
- The human body (produced naturally during digestion)
- Dairy products like yogurt and cheese
- Fermented foods like kimchi and sourdough bread
It's a natural substance, but that doesn't make concentrated forms any less hazardous.
Common Uses of Acetic Acid
Acetic acid turns up in more places than most people expect, from factory floors to medicine cabinets. Products that contain acetic acid use different concentrations, which can be found when looking up its Safety Data Sheet (SDS).
Industrial and Manufacturing
The biggest industrial use of acetic acid is making vinyl acetate, a building block for paints, glues, and coatings. It's also used to make acetic anhydride, which is used to produce synthetic fabrics and photographic film materials.
Pharmaceutical and Medical
In medicine, acetic acid is used to produce vitamins, antibiotics, and certain hormones. Diluted solutions are used to treat ear infections, and in some parts of the world, it's used as a low-cost screening tool for cervical cancer.
Food Industry
As a food additive (labeled E260), acetic acid acts as a preservative and helps control acidity. It's found in pickles, condiments, and many processed foods.
Household Cleaning
White vinegar, a diluted form of acetic acid, is a popular household cleaner. It removes mineral buildup, cuts through grease, and can disinfect many surfaces.
Textile and Leather
In fabric production, acetic acid helps regulate pH during the dyeing process, which makes colors absorb into materials more evenly.
Acetic Acid Dangers: What You Need to Know
How dangerous acetic acid is mostly depends on its strength (how concentrated it is). A splash of vinegar won't hurt you, but exposure to glacial or industrial-strength acetic acid is a much more serious risk. The dangers of acetic acid vary depending on the route of exposure, which include:
Skin and Eye Contact
Concentrated acetic acid is highly corrosive. Skin contact can cause severe chemical burns, and even short exposure to high concentrations can damage tissue. Eye contact is especially dangerous and can lead to permanent injury if not treated right away.
Breathing It In
Breathing in acetic acid vapors irritates the nose, throat, and airways. Heavy or long-term exposure can cause swelling of the throat, chronic bronchitis, and lung damage.
OSHA sets the maximum safe exposure at 10 ppm over an 8-hour workday, with a short-term limit of 15 ppm. NIOSH considers anything above 50 ppm immediately dangerous to life and health.
Swallowing It
Swallowing acetic acid, even in moderate amounts, can burn the mouth, throat, and stomach. Internal damage from ingestion may not show up right away, which makes it especially dangerous.
Fire and Explosion Risk
Acetic acid catches fire relatively easily, with a flash point of around 39°C (102°F). Its vapors can form explosive mixtures in the air when heated. Keep it away from open flames, sparks, and hot surfaces.
Chemical Reactions
Acetic acid reacts badly with strong oxidizers like nitric acid, chromic acid, and sodium peroxide, as well as with strong bases and certain metals. These reactions can produce toxic gases or start fires.
How to Handle Acetic Acid Safely
Whether you're working with it in a lab, a factory, or using a cleaning product that contains it, the same basic principles apply.
Wear the Right PPE
When working with concentrated acetic acid, always use:
- Chemical-resistant gloves (nitrile or neoprene work well)
- Safety goggles or a face shield to keep splashes away from your eyes
- A lab coat or chemical-resistant apron
- A respirator in areas with poor airflow (look for a NIOSH-approved organic vapor respirator for concentrations above 10 ppm)
Make Sure the Area Is Ventilated
Always work with acetic acid in a well-ventilated space, or under a fume hood in a lab. Glacial acetic acid fumes build up quickly in closed spaces, so good airflow isn't optional.
Store It Properly
Keep acetic acid in a cool, dry, well-ventilated area, away from heat sources, open flames, and chemicals it reacts badly with (oxidizers and strong bases especially). Keep containers sealed tight and never store it near food or drinks.
Handling a Spill
If there is a spill, clear the area and get air moving through the space. Use a chemical spill kit made for corrosive liquids. Don't let it wash into drains or waterways since it can reduce oxygen levels in water and harm aquatic life.
First Aid
The first aid that needs to be applied depends on your route of exposure, which includes:
- Skin contact: Flush with water for at least 15 minutes. Take off any clothing that got splashed. See a doctor.
- Eye contact: Remove contact lenses right away. Rinse eyes with water for at least 15 minutes. Get emergency medical help immediately.
- Inhaled vapors: Get to fresh air right away. If breathing is difficult, give oxygen and call emergency services.
- Swallowing: Don’t induce vomiting. Rinse mouth with water and call Poison Control or a doctor immediately.
Final Thoughts
Acetic acid is everywhere, from the vinegar on your shelf to the coatings on industrial equipment. At low concentrations, it's safe and useful. At high concentrations, it's a serious chemical hazard that deserves proper training, the right protective gear, and careful storage.
If you work somewhere that uses acetic acid, your workplace should have a current Safety Data Sheet (SDS) on file, proper employee training in place, and a clear plan for handling emergencies. That's not a suggestion. It's a basic part of keeping people safe on the job.
Frequently Asked Questions
Is acetic acid the same as vinegar?
Not exactly. Vinegar is a diluted mix that's 5-8% acetic acid in water. Pure acetic acid is far more concentrated and much more hazardous than what you'd find in your kitchen.
What are the most common symptoms of acetic acid exposure?
Common symptoms include irritation of the eyes, nose, and throat, skin burns, coughing, and trouble breathing. High concentrations can cause severe burns and respiratory damage.
Is it safe to use acetic acid at home?
At household concentrations found in vinegar and cleaning products, yes. Keep it away from children, don't mix it with bleach or strong bases, and make sure the area is ventilated when using it.
What is glacial acetic acid used for?
It's the industrial form of acetic acid, used to make vinyl acetate, acetic anhydride, pharmaceuticals, and chemical solvents. It's not a consumer product and requires strict handling precautions.
What PPE do you need when handling concentrated acetic acid?
At a minimum, you need chemical-resistant gloves, splash-proof goggles, and a lab coat or apron. In poorly ventilated spaces, add a NIOSH-approved organic vapor respirator and a full face shield.

