

The EU's 22nd ATP to the CLP Regulation takes effect on 1 May 2026. With 50 chemical entry changes now mandatory, here is a full breakdown of what changed, which substances are affected, and what the wider regulatory picture looks like heading into the second half of the year.
The European Commission adopted the 22nd Adaptation to Technical Progress (ATP) to the Classification, Labelling and Packaging (CLP) Regulation (EC No 1272/2008).
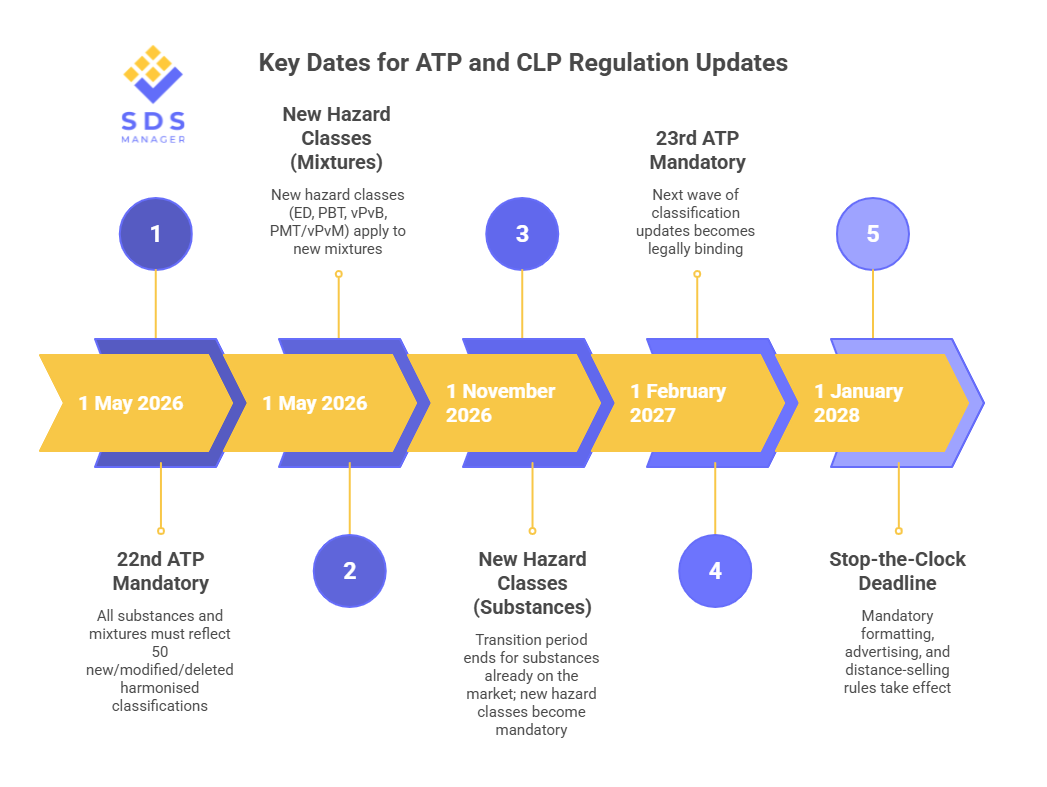
Key dates to know:
- 19 June 2024 — Adopted by the European Commission
- 30 September 2024 — Published in the Official Journal of the European Union
- 20 October 2024 — Entered into force
- 1 May 2026 — Mandatory across all EU member states
The Numbers Behind the 22nd ATP
| Change Type | Substance / Group | CAS No. | Key Classification / Change |
|---|---|---|---|
| New | Helional | 1205-17-0 | Repr. 1B |
| New | 2,4-dimethylcyclohex-3-ene-1-carbaldehyde | 68039-49-6 | Repr. 1B |
| New | Hexyl salicylate | 6259-76-3 | Repr. 2; Skin Sens. 1 |
| New | Acetone oxime | 127-06-0 | CMR Cat. 1 (professional use only) |
| New | Sodium 3-(allyloxy)-2-hydroxypropanesulphonate | 52556-42-0 | CMR Cat. 1 |
| New | Benthiavalicarb-isopropyl | 177406-68-7 | CMR Cat. 1 |
| New | Silver (massive, powder, nano forms) | 7440-22-4 | Repr. 2; nano also STOT RE 2 + Aquatic toxicity |
| New | Copper powder/flakes | 7440-50-8 | Aquatic Acute 1; Aquatic Chronic 1 |
| New | Multi-walled carbon nanotubes | — | Carc. 1B; STOT RE 1 (lungs) |
| New | Benzotriazole & salts | 95-14-7 | Aquatic Chronic 2 |
| New | Sulfur | 7704-34-9 | Aquatic Chronic 2 |
| New | Glyphosate | 1071-83-6 | Aquatic Chronic 2 |
| New | S-metolachlor | 87392-12-9 | Aquatic Chronic 1 |
| New | Pyraclostrobin | 175013-18-0 | Aquatic Chronic 1 |
| New | Fenpropidin | 67306-00-7 | Aquatic Chronic 1 |
| New | 2,3-Epoxypropyl neodecanoate | 26761-45-5 | Repr. 1B; Skin Sens. 1 |
| New | o-Phenylphenol | 90-43-7 | Carc. 2 |
| New | Perboric acid / sodium perborate salts | Various | Repr. 1B; SCL removed; ATE added |
| New | Trimethyl borate | 121-43-7 | Repr. 1B |
| New | 2-Methylnaphthalene | 91-57-6 | Aquatic Chronic 1 |
| New | Chlorothalonil | 1897-45-6 | Aquatic Chronic 1 |
| New | Flurochloridone | 61213-25-0 | Aquatic Chronic 1 |
| Modified | n-Hexane | 110-54-3 | Upgraded to STOT RE 1 |
| Modified | Formaldehyde | 50-00-0 | Reclassification + Skin Sens. 1 added |
| Modified | Formic acid | 64-18-6 | Acute toxicity added |
| Modified | Peracetic acid | 79-21-0 | Environmental toxicity expanded |
| Modified | Dibenzoyl peroxide | 94-36-0 | Environmental toxicity expanded |
| Modified | Dicamba | 1918-00-9 | Environmental toxicity updated |
| Modified | Glyphosate, Pyraclostrobin, Fenpropidin, S-metolachlor, Sulfur | Various | Environmental classifications updated |
| Modified | Copper compounds | Various | Aquatic Acute/Chronic classifications expanded |
| Deleted | Perborate / borate sodium salts (multiple index numbers) | Various | Entries removed |
| Deleted | Copper compound entry (granulated copper) | — | Replaced by new surface-area-specific entry |
The 22nd ATP amends Table 3 of Part 3 of Annex VI to the CLP Regulation with the following changes:
- 27 new harmonised classifications added
- 16 existing entries modified
- 7 entries deleted
That is 50 total changes to the harmonised classification list.
Which Substances Are Affected
Modified entries: tightened classifications
N-hexane and formaldehyde
Two of the most common solvents in industrial use, have received more stringent classifications due to updated human health assessments. Formic acid has been revised based on the latest physical property data.
Trimethyl borate
It received one of the more significant upgrades in this ATP, reclassified as Reprotoxic Category 1B, a meaningful step up from its previous status.
Peracetic acid and dibenzoyl peroxide
They now carry expanded classifications covering both acute and chronic environmental toxicity.
Dicamba and glyphosate
Two active substances that have attracted sustained scientific and political debate across the EU, both receive revised harmonised classifications extending into environmental hazard categories.
New entries added
Silver
Across massive, powder, and nano forms received harmonised classification for the first time. The Commission settled on Reproductive Toxicity Category 2 (H361f), a less stringent outcome than the Category 1 classification proposed during the consultation process.
Copper (specific surface area > 0.67 mm²/mg)
Replaced the previous granulated copper entry and is now classified as Aquatic Acute 1 and Aquatic Chronic 1, carrying an M-Factor of 10 for acute aquatic toxicity.
Hexyl salicylate
Which is widely used in laundry products, air fresheners, and the broader fragrance industry, is added to the harmonised list for the first time. Several CMR Category 1 substances have also been newly classified with restrictions limiting them to professional use only, including acetone oxime.
Deleted entries
Seven entries have been removed from Annex VI, including several borate index numbers that have been consolidated or superseded by updated entries elsewhere in the table.
What the 1 May 2026 Deadline Requires
From 1 May 2026, all substances and mixtures placed on the EU market must be classified, labelled, and packaged in line with the 22nd ATP.
The obligations this triggers cover several areas. Labels on affected products must carry revised hazard statements, pictograms, and precautionary information reflecting the updated classifications.
Safety Data Sheets require updates across multiple sections, most commonly:
- Section 2 — Hazard Identification
- Section 3 — Composition and Ingredients
- Section 8 — Exposure Controls and Personal Protection
- Section 15 — Regulatory Information
Classification and Labelling notifications must be submitted or updated in ECHA's C&L Inventory, and under CLP's information-in-the-supply-chain requirements, updated hazard data must flow downstream to customers and distributors without delay.
Geography offers no exemption. Any company shipping chemical products into the EU, wherever it is based, falls within the scope of the 22nd ATP. Compliant documentation reflecting the 22nd ATP classifications is a prerequisite for continued market access from 1 May 2026 onwards. The 22nd ATP applies only in the EU. The UK maintains its own independent GB CLP mandatory classification list through a separate process.
The Wider 2026 Regulatory Picture
The 22nd ATP does not exist in isolation. Companies tracking EU chemical compliance in 2026 are managing a dense and overlapping set of deadlines.
The 23rd ATP has already been adopted and will apply from 1 February 2027, meaning the classification update cycle continues without pause. ECHA's Annex VI table shows both ATPs are already published and available for early adoption.
The postponed provisions cover mandatory label formatting rules, advertising requirements, online and distance selling obligations, and fuel pump labelling. The new hazard class classification requirements and the 22nd ATP deadline of 1 May 2026 are not affected by this postponement and remain on schedule.
The Commission estimates the broader simplification package, part of the so-called Omnibus VI, could save the chemical industry at least €363 million annually. The package sits within the EU's wider regulatory simplification drive, framed partly around the Draghi competitiveness report and the Budapest Declaration's call for a "simplification revolution."

